- Home
-
Screening
- Ionic Screening Service
-
Ionic Screening Panel
- Ligand Gated Ion Channels
- Glycine Receptors
- 5-HT Receptors3
- Nicotinic Acetylcholine Receptors
- Ionotropic Glutamate-gated Receptors
- GABAa Receptors
- Cystic Fibrosis Transmembrane Conductance Regulators (CFTR)
- ATP gated P2X Channels
- Voltage-Gated Ion Channels
- Calcium Channels
- Chloride Channels
- Potassium Channels
- Sodium Channels
- ASICs
- TRP Channels
- Other Ion Channels
- Stable Cell Lines
- Cardiology
- Neurology
- Ophthalmology
-
Platform
-
Experiment Systems
- Xenopus Oocyte Screening Model
- Acute Isolated Cardiomyocytes
- Acute Dissociated Neurons
- Primary Cultured Neurons
- Cultured Neuronal Cell Lines
- iPSC-derived Cardiomyocytes/Neurons
- Acute/Cultured Organotypic Brain Slices
- Oxygen Glucose Deprivation Model
- 3D Cell Culture
- iPSC-derived Neurons
- Isolation and culture of neural stem/progenitor cells
- Animal Models
- Techinques
- Resource
- Equipment
-
Experiment Systems
- Order
- Careers
3D Cardiotoxicity
Interspecies differences in ion channel composition and signal cascades can hinder the predictivity of the cardio-toxicity test by using non-human experimental models. However, traditional 2D cell culture models cannot recapitulate the human in vivo structure and organization of the myocardium and are lack of the crucial process of cardiomyocyte differentiation. What's more, conventional static 2D culture of adult myocytes fails to preserve the adult phenotype.
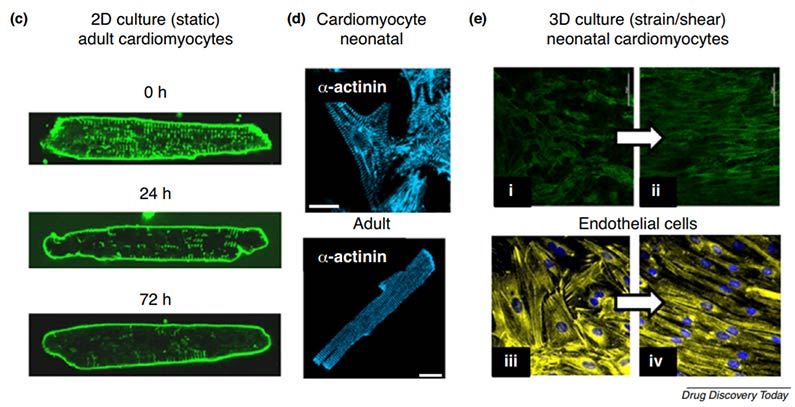
Fig. 1 Importance of the microenvironment to cardiovascular cellular phenotypes
To overcome the weaknesses and limitations of current cell-based and animal-based testing, 3D models with the appropriate biophysical tissue architecture are generated for cardio-toxicity test in order to provide highly predictive and detailed information for the human in vivo study. Creative Bioarray offers the opportunity for highly predictable pre-clinical cardio-toxicity testing to help our customers to reduce both the cost and duration of bringing a new drug candidate to market.
Advantages
3D structure of cardiac cell models can be more representative of in vivo tissue than traditional monolayer cell culture
Capable of providing several physiologic model systems of 3D culture made by cell lines, human iPS cells and ES cells for you to choose
Able to cover most test endpoints for cardio-toxicity test
Applications
Creative Bioarray tests your target compounds with series concentrations. We focus on a wide range of test endpoints include: QT interval, beat rate change, contraction rate and duration as well as the compound impact on different ion channels, which allow you to better characterize your compounds.
Work flow
Experiment Example
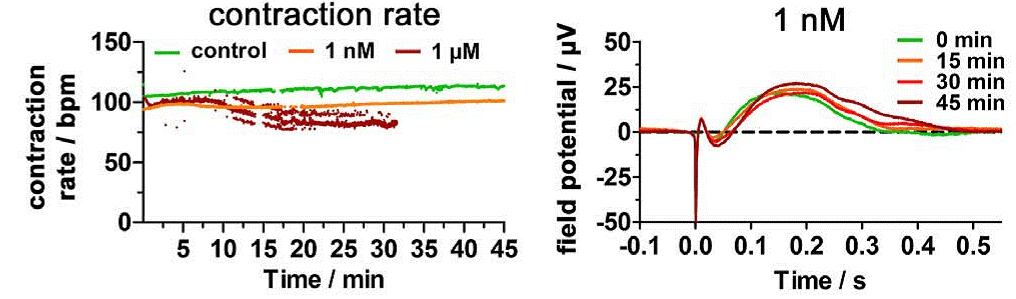
Fig. 2 The representative data analysis for the detection of APD-prolongation effects of compound E4031
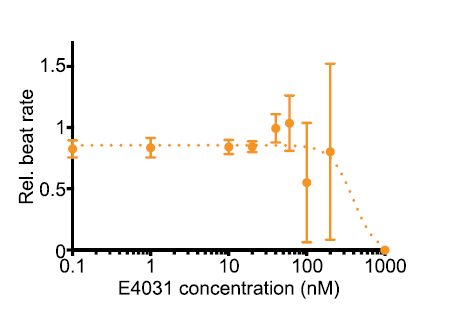
Fig. 3 The representative data analysis for the dose-dependent decrease in beat rate of E4031 treatment
Quotation and Ordering
If you have any special needs in our 3D Cardiotoxicity Service, please contact us at Email or Telephone for this special service. Let us know what you need and we will accommodate you. We look forward to working with you in the future.
References
Barros-Gomes, S., et al. Rationale for setting up a cardio-oncology unit: our experience at Mayo Clinic. Cardio-Oncology. 2016; 2: 5.
Ryan AJ, et al. Towards 3D in vitro models for the study of cardiovascular tissues and disease. Drug Discov Today 2016; 21: 1437–1445.
Mathur A, et al. Human iPSC-based cardiac microphysiological system for drug screening applications. Sci Rep 2015; 5: 8883.
Jahnke H-G, et al. A novel 3D label-free monitoring system of hES-derived cardiomyocyte clusters: a step forward to in vitro cardiotoxicity testing. PLoS One 2013; 8: e68971.
Related Section
Inquiry

