- Home
-
Screening
- Ionic Screening Service
-
Ionic Screening Panel
- Ligand Gated Ion Channels
- Glycine Receptors
- 5-HT Receptors3
- Nicotinic Acetylcholine Receptors
- Ionotropic Glutamate-gated Receptors
- GABAa Receptors
- Cystic Fibrosis Transmembrane Conductance Regulators (CFTR)
- ATP gated P2X Channels
- Voltage-Gated Ion Channels
- Calcium Channels
- Chloride Channels
- Potassium Channels
- Sodium Channels
- ASICs
- TRP Channels
- Other Ion Channels
- Stable Cell Lines
- Cardiology
- Neurology
- Ophthalmology
-
Platform
-
Experiment Systems
- Xenopus Oocyte Screening Model
- Acute Isolated Cardiomyocytes
- Acute Dissociated Neurons
- Primary Cultured Neurons
- Cultured Neuronal Cell Lines
- iPSC-derived Cardiomyocytes/Neurons
- Acute/Cultured Organotypic Brain Slices
- Oxygen Glucose Deprivation Model
- 3D Cell Culture
- iPSC-derived Neurons
- Isolation and culture of neural stem/progenitor cells
- Animal Models
- Techinques
- Resource
- Equipment
-
Experiment Systems
- Order
- Careers
Alzheimer's Disease Model
Alzheimer's disease (AD) is the most prevalent neurodegenerative disorder in the world which causes dementia and affects about 36 million people worldwide. The pathogenesis is still unclear, although may support the Amyloid cascade theory, which is based on abnormal APP processing and clearance, leading to the formation of toxic amyloid oligomers, fibrils and extracellular amyloid plaques. Amyloid seems to trigger the tau protein dependent neurofibrillary pathology, which is another pathological hallmark of AD. The whole process is long-lasting and may start decades before the first disease symptoms occur. In spite of enormous research efforts, so far no efficient treatment is available. For successful drug development, validated predictive animal models are needed. Animal models mimicking human Alzheimer's Disease allow us to test therapeutic potentials and investigate disease progression, contributing to improvement of quality of life for humans.
Creative Bioarray currently offers research with several different APP transgenic rodent models which are different in the time course of development of onset of Abeta-pathology, progression pattern, neuro-inflammation and cognitive deficits. In addition, an advanced model of tau-pathology is available. In these mice a tetracycline responsive element allows control of tau expression (TET-off).
Whether it is in vitro or in vivo electrophysiological approaches, whole animal behavior or molecular biology techniques, our research teams will provide expert consultancy in putting together the study proposal you require, and our investigators will then rapidly deliver data of the highest quality to drive forward your research programs.

Genetically modified rodent models, expressing human (wild type or mutated) APP or human Tau (wildtype or mutated) replicate aspects of the pathogenetic cascade and provide useful tools for assessing the therapeutic potential of new treatment strategies.
The value of these models is critically depending on the experience and skills of the investigators, strictly following principles of good scientific practice. Creative Bioarray provides fully blinded randomized studies, based on power calculation. Creative Bioarray has extensive experience in delivering first class preclinical translational research into the study, treatment and prevention of neurodegenerative disorders, particularly around amyloid based animal models of Alzheimer's disease.
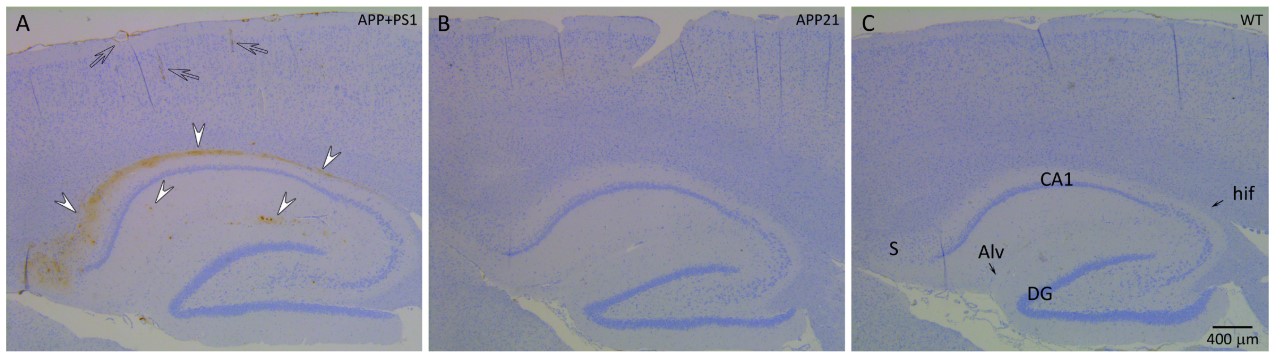
Fig. 1 Immunohistochemistry of the hippocampus and the cortex of an APP+PS1 (A), APP21 (B) and WT (C) rat using 6E10 monoclonal antibody raised against Aβ. Brown staining indicates the presence of Aβ.
Requirements
In order to properly examine the disease, ideal animal models of AD would need to exhibit these;
important attributes;
Developing Aβ plaques and CAA;
Predominantly producing high-dose Aβ 42;
Promoting Aβ oligomer formation;
Producing intraneuronal accumulation of Aβ;
Inducing hyperphosphorylation of tau and developing intraneuronal tangles;
Enhancing neurodegeneration with synaptic loss, axonal damage, and defects in neurogenesis; Showing behavioral (learning and memory) and electrophysiological (LTP, synaptic plasticity) impairments that correspond to the pattern of neurodegeneration.
Since the first murine model of AD was reported one and a half decades ago, many other models have been generated by modification of transgenes that cause familial AD. These models have contributed to significant basic research and numerous therapeutic innovations to treat AD and halt disease progression. Although most of the models mirror featured aspects of AD pathogenesis such as β -amyloidosis, NFT formation, cognitive dysfunction, and/or synaptic loss, no perfect model matching human AD is currently available.
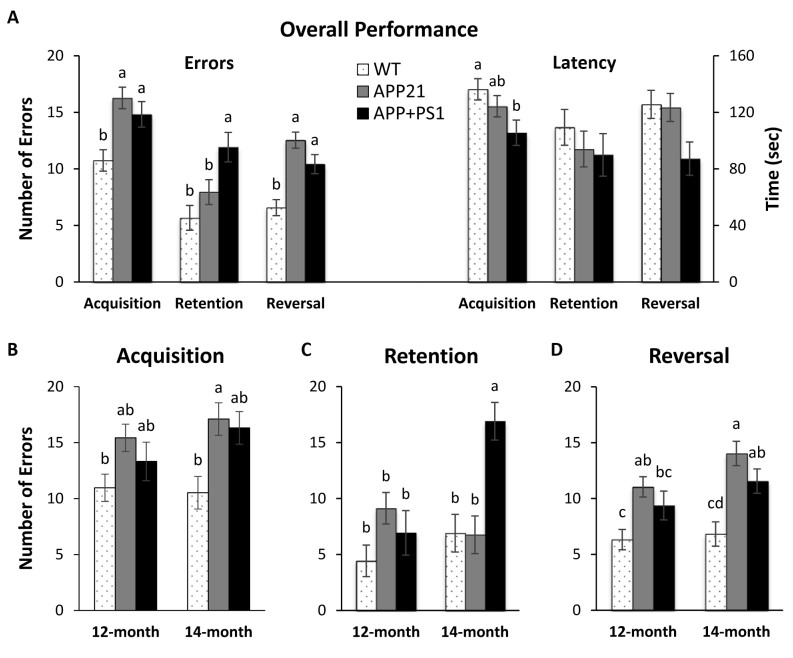
Fig. 2 Barnes Maze results
Therefore, it is necessary to choose an animal model of AD that is appropriate and best fit for the study- specific aims, symptom components, and outcomes. The most popular animal models of AD are Tg mice overexpressing APP, PS-1, and/or PS-2 with single or multiple FAD-linked mutations. APP Tg and APP/PS double-Tg mice exhibit key features of AD such as high levels of Aβ and formation of Aβ deposition, but NFTs are not generated. Therefore, mouse models overexpressing tau or a mutant form of tau with NFT formation are utilized to investigate the role of tau dysfunction and NFTs. Although severe neuronal loss is not found in any current mouse model, impaired neurogenesis is observed in some of the models.
Since no model perfectly matching human AD is currently available, it is important to choose an appropriate model to fit a specific research target. Creative Bioarray has experienced experts to work with you to choose and develop the most appropriate Alzheimer's Disease model for your studies, and help you set up a detailed study plan.
Reference
Klakotskaia D, et al. Memory deficiency, cerebral amyloid angiopathy, and amyloid-β plaques in APP+PS1 double transgenic rat model of Alzheimer's disease. PLoS One. 2018; 13: 1–16.
Related Section
- Parkinson's Disease Model
- Huntington's Disease Model
- Epilepsy Model
- ALS Model
- Psychiatric Model
- Autism Spectrum Disorder
- Cerebral-Spinal Injury Model
- Pain Model
- Stroke Model
- Fragile X Model
- Acute/Chronic Heart Failure Model
Inquiry

