- Home
-
Screening
- Ionic Screening Service
-
Ionic Screening Panel
- Ligand Gated Ion Channels
- Glycine Receptors
- 5-HT Receptors3
- Nicotinic Acetylcholine Receptors
- Ionotropic Glutamate-gated Receptors
- GABAa Receptors
- Cystic Fibrosis Transmembrane Conductance Regulators (CFTR)
- ATP gated P2X Channels
- Voltage-Gated Ion Channels
- Calcium Channels
- Chloride Channels
- Potassium Channels
- Sodium Channels
- ASICs
- TRP Channels
- Other Ion Channels
- Stable Cell Lines
- Cardiology
- Neurology
- Ophthalmology
-
Platform
-
Experiment Systems
- Xenopus Oocyte Screening Model
- Acute Isolated Cardiomyocytes
- Acute Dissociated Neurons
- Primary Cultured Neurons
- Cultured Neuronal Cell Lines
- iPSC-derived Cardiomyocytes/Neurons
- Acute/Cultured Organotypic Brain Slices
- Oxygen Glucose Deprivation Model
- 3D Cell Culture
- iPSC-derived Neurons
- Isolation and culture of neural stem/progenitor cells
- Animal Models
- Techinques
- Resource
- Equipment
-
Experiment Systems
- Order
- Careers
ALS Model
Amyotrophic Lateral Sclerosis (ALS) is a late adult-onset, quickly progressing paralytic disorder marked by selective degeneration of motor neurons in the motor cortex and spinal cord. For ALS patients, this disease leads to paralysis and eventual death. ALS patients are classified into two main categories, sporadic (sALS) and familial (fALS). Both fALS and sALS patients present with muscle weakness and atrophy, as well as spasticity due to the loss of both upper and lower motor neurons.
A rather large subset (~20%) of fALS cases have been linked to mutations in the Cu/Zn superoxide dismutase 1 gene (SOD1). Earliest models used toxic levels or deficiencies of metals such as lead or mercury as a way to study the disease. Genetic mutations in superoxide dis-mutase 1 (SOD1), fused in sarcoma/translocated in liposarcoma (FUS/TLS), and transactivation response (TAR) DNA-binding protein with molecular weight 43 kDa (TDP - 43) genes have been identified in 25–35% of the fALS cases. Due to numerous possible etiologies of sALS, most of the current research models focus on genetic etiologies. Still, the majority of early research was performed using observational or spontaneous cases of ALS in both humans and animals, until technology and knowledge allowed more complex models with genetic manipulation.
Creative Bioarray fully understands advantages and limitations between each kind of animal models for investigating ALS. It is essential for your research to find the best matching animal model at the beginning. We will work side-by-side with you to decide the most appropriate model for your specific experimental hypotheses.
Current research into neurodegenerative disorders relies heavily on animal models that recapitulate human disease. A large number of animal models are toxin-based, utilizing chemicals or metals that have been correlated with human disease. As a whole, current animal models serve as adequate substitutes to their human counterparts, providing valuable tools for translational and basic science research in ALS, with the caveat that most models typically do not recapitulate the totality of the human disease.
For many years, the gold standard for modeling fALS has been murine models of human SOD1 mutations; however, more recent models have been developed which may better recapitulate different aspects of ALS disease progression. Nevertheless, even current models may not fully mimic all facets of disease etiology, progression, or pathology, necessitating continued research into developing models that more closely replicate the clinical manifestations of the disease.
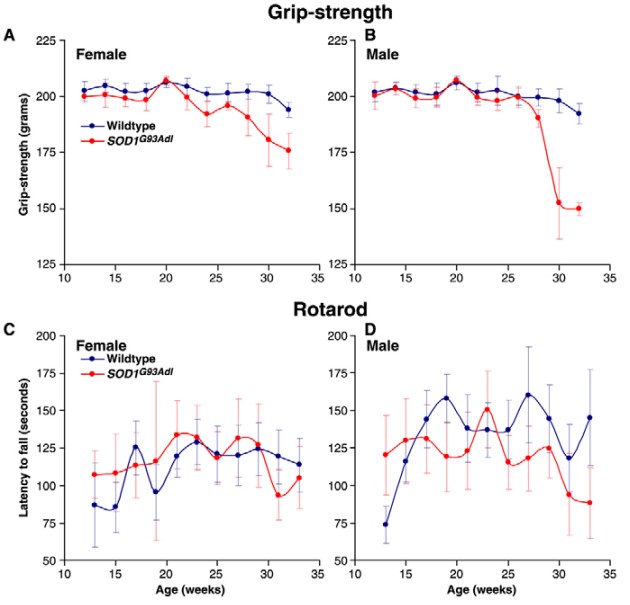
Fig. 1 Grip-strength and rotarod analysis of SOD1G93Adl B6 mice.
For ALS, genetic models have been generated based on the clinical reports of familial forms of the disease. Mutations in genes coding for SOD1, FUS/TLS, and TDP-43 have been used for ALS models. Transgenic mice expressing the mutant form of human SOD1 become paralyzed as a result of neuronal loss from the brain and spinal cord and die at 5-6 months of age. These mutant mice show a phenotype typical of motor neuron loss which includes severe muscle wasting and hindlimb paralysis. Recent evidence suggests that the appearance of symptoms is associated with an accumulation of detergent-insoluble, sedimentable, forms of mutant SOD1 in spinal cords and brainstems.
The availability of this mutant mouse model has advanced our understanding of the mechanisms involved in ALS, while at the same time supported the development of novel drug treatments in this disease category. Creative Bioarray offers a large panel of tests using the most widely chosen mouse model of ALS. These tests include standard motor function measures such as grip strength and rotarod as well as more advances computer vision metrics for gait measures and whole animal behavior analysis.
Creative Bioarray is dedicated to create the most suitable animal models of ALS to advance the development for potential therapeutics into clinical studies. We will be with you on every step for obtaining meaningful and suggesting data from animal model studies for ALS.
Reference
Acevedo-Arozena A, et al. A comprehensive assessment of the SOD1G93A low-copy transgenic mouse, which models human amyotrophic lateral sclerosis. Dis Model Mech. 2011; 4: 686–700.
Related Section
- Alzheimer's Disease Model
- Parkinson's Disease Model
- Huntington's Disease Model
- Epilepsy Model
- Psychiatric Model
- Autism Spectrum Disorder
- Cerebral-Spinal Injury Model
- Pain Model
- Stroke Model
- Fragile X Model
- Acute/Chronic Heart Failure Model
Inquiry

