- Home
-
Screening
- Ionic Screening Service
-
Ionic Screening Panel
- Ligand Gated Ion Channels
- Glycine Receptors
- 5-HT Receptors3
- Nicotinic Acetylcholine Receptors
- Ionotropic Glutamate-gated Receptors
- GABAa Receptors
- Cystic Fibrosis Transmembrane Conductance Regulators (CFTR)
- ATP gated P2X Channels
- Voltage-Gated Ion Channels
- Calcium Channels
- Chloride Channels
- Potassium Channels
- Sodium Channels
- ASICs
- TRP Channels
- Other Ion Channels
- Stable Cell Lines
- Cardiology
- Neurology
- Ophthalmology
-
Platform
-
Experiment Systems
- Xenopus Oocyte Screening Model
- Acute Isolated Cardiomyocytes
- Acute Dissociated Neurons
- Primary Cultured Neurons
- Cultured Neuronal Cell Lines
- iPSC-derived Cardiomyocytes/Neurons
- Acute/Cultured Organotypic Brain Slices
- Oxygen Glucose Deprivation Model
- 3D Cell Culture
- iPSC-derived Neurons
- Isolation and culture of neural stem/progenitor cells
- Animal Models
- Techinques
- Resource
- Equipment
-
Experiment Systems
- Order
- Careers
Parkinson's Disease Model
Parkinson's disease (PD) is currently the second most common age-related neurodegenerative disease after Alzheimer's disease, mainly affecting the motor system, resulting in progressive death of dopamine-generating cells in the substantia nigra pars compacta (SNpc). Beside the most obvious symptoms, movement-related symptoms such as shaking, rigidity, slowness of movement and gait, patients develop non-motor symptoms, afflicting personality and behavior (depression, anxiety, apathy), as well disturbances in cognitive function.
The etio-pathogenesis of the disease is not fully explored as the majority of patient cases are sporadic; however, mutations in alpha-synuclein causing abnormally aggregated ("Lewy bodies"), and mitochondrial genes like Pink-1 or DJ-1 have been linked to the pathology. Certain toxin exposure also increases (different pesticides e.g. rotenone) the risk. Genetic and chemical induced lesion disease models are available to study the disease mechanisms and for drug screening.
Creative Bioarray is dedicated to create the most suitable animal models of Parkinson's disease to advance the development for potential therapeutics into clinical studies. We will be with you on every step for obtaining meaningful and suggesting data from animal model studies for Parkinson's disease.

While the disease can be accurately diagnosed, and symptoms can be alleviated using levo-(l)-3,4-dihydroxyphenylalanine (L-DOPA), current therapeutics can neither cure the disorder nor completely stop disease progression.
Animal models have been created that assess pathogenesis resulting from genetic modifications, as well as neurotoxic insult. Creative Bioarray provides one of the most comprehensive service platforms for animal model-based Parkinson's disease studies, from model selection, model creation to therapeutics assessment, so that to accelerate the advancement for new drug and therapeutics from pre-clinical into clinical studies.
Classical models for Parkinson's disease are toxin-based animal models, the unilateral 6-OHDA-lesioned rodent and the MPTP-treated primate. More extended animal models including transgenic animals like those related to PINK1, Parkin, alpha-synuclein and LRRK2 alterations. And other vertebrate models such as zebrafish and invertebrate models like fruit fly and round worms are ideal for studying the molecular and cellular mechanisms of Parkinson's disease. Creative Bioarray fully understands advantages and limitations between each kind of animal models for investigating Parkinson's disease. It is essential for your research to find the best matching animal model at the beginning. We will work side-by-side with you to decide the most appropriate model for your specific experimental hypotheses.
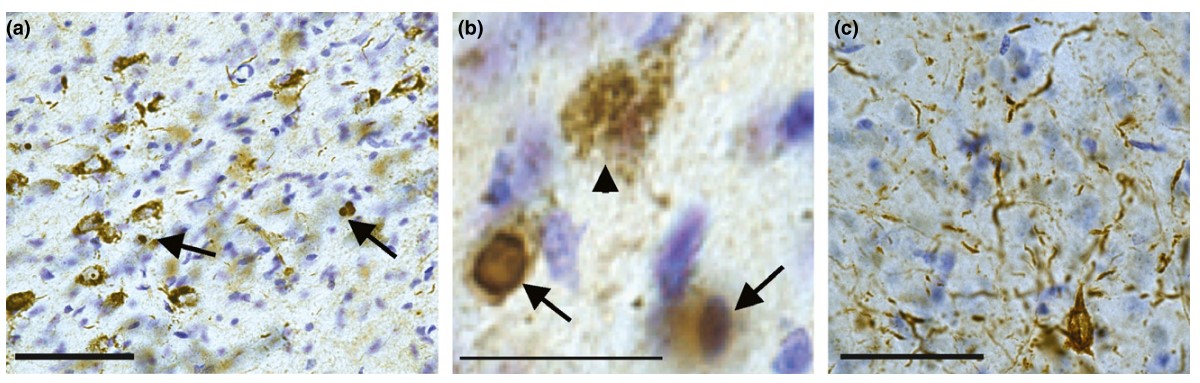
Fig. 1 Injections of fibrils into the substantia nigra pars compacta induces the formation of pS129- -synuclein inclusions with morphologies similar to Lewy bodies and Lewy neurites found in PD.
Advantages
Our strong capacity includes, but is not limited to:
Various animal model selections: mice, rats, and Non-Human Primates.
Local access to large NHP resource (>120,000 population).
Up-to-date facility and medical equipment, such as PET scan and MRI.
Well-trained surgical and medical imaging teams.
Talented and inspiration Ph.D level scientists in neurodegeneration diseases models.
Models
Our Parkinson's disease models include, but are not limited to:
Unilateral or Systemical Parkinsonism
L-Dopa induced dyskinesis
Mild Cognition Impairment (MCI) complication
We have experienced experts to work with you to choose and develop the most appropriate model for your studies, and help you set up a detailed study plan. Furthermore, we also assist you with project executing, data validation and final report.
Services
Our one-stop full custom Parkinson's disease model service includes, but is not limited to:
Model creation
Behavior assessment
Comprehensive imaging analysis
Lab analysis: biochemical and routine parameters, quantitative analysis of proteins, and histopathology testings.
Reference
Volpicelli-Daley LA, et al. How can rAAV-α-synuclein and the fibril α-synuclein models advance our understanding of Parkinson's disease? J Neurochem. 2016; 139: 131–155.
Related Section
- Alzheimer's Disease Model
- Huntington's Disease Model
- Epilepsy Model
- ALS Model
- Psychiatric Model
- Autism Spectrum Disorder
- Cerebral-Spinal Injury Model
- Pain Model
- Stroke Model
- Fragile X Model
- Acute/Chronic Heart Failure Model
Inquiry

