- Home
-
Screening
- Ionic Screening Service
-
Ionic Screening Panel
- Ligand Gated Ion Channels
- Glycine Receptors
- 5-HT Receptors3
- Nicotinic Acetylcholine Receptors
- Ionotropic Glutamate-gated Receptors
- GABAa Receptors
- Cystic Fibrosis Transmembrane Conductance Regulators (CFTR)
- ATP gated P2X Channels
- Voltage-Gated Ion Channels
- Calcium Channels
- Chloride Channels
- Potassium Channels
- Sodium Channels
- ASICs
- TRP Channels
- Other Ion Channels
- Stable Cell Lines
- Cardiology
- Neurology
- Ophthalmology
-
Platform
-
Experiment Systems
- Xenopus Oocyte Screening Model
- Acute Isolated Cardiomyocytes
- Acute Dissociated Neurons
- Primary Cultured Neurons
- Cultured Neuronal Cell Lines
- iPSC-derived Cardiomyocytes/Neurons
- Acute/Cultured Organotypic Brain Slices
- Oxygen Glucose Deprivation Model
- 3D Cell Culture
- iPSC-derived Neurons
- Isolation and culture of neural stem/progenitor cells
- Animal Models
- Techinques
- Resource
- Equipment
-
Experiment Systems
- Order
- Careers
QT Service Using Langendorff Perfused Heart
As the most common reason for cardiotoxicity, QT interval prolongation can serve as a direct indicator for drug cardiotoxicity. The QT interval, a measure of time from the start of the Q wave to the end of the T wave, represents the electrical depolarization and repolarization of the ventricles. QT prolongation implies a delayed ventricular repolarization which may cause ventricular tachyarrhythmias and possibly sudden cardiac death. Among all drug safety issues, QT prolongation is one of the leading causes of therapeutic drug failures during preclinical development.
Creative Bioarray provides QT prolongation assay using isolated heart (Langendorff Assay) to evaluate the cardiotoxicity of compounds with QT interval as the indicator. The potential to prolong the QT interval has a major impact on drug development programs. A rapid, inexpensive, reproducible and sensitive predictions of QT-prolonging potential can be provided by isolated heart preparation by Langendorff preparation. Moreover, this preparation allows the assessment of the effects of a drug on electrophysiological (ECG, MAP) and mechanical (left ventricular tension, coronary pressure and flow rate) parameters, yielding a variety of safety or toxicity indicators in a single study. Common disease phenotypes are easily reproduced using this complex ex vivo model.
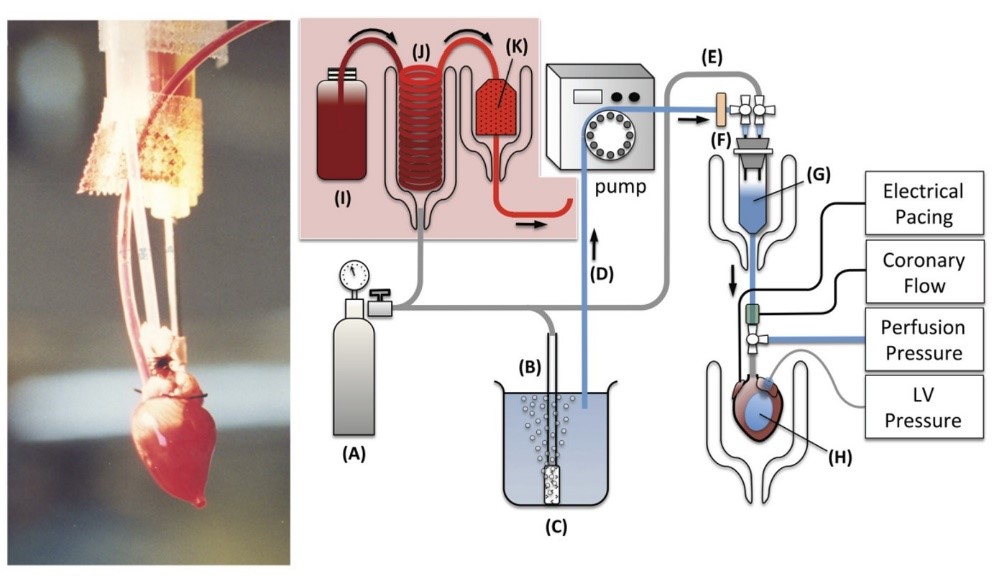
Fig. 1 isolated isovolumically contracting mouse heart erythrocyte perfused in the Langendorff mode
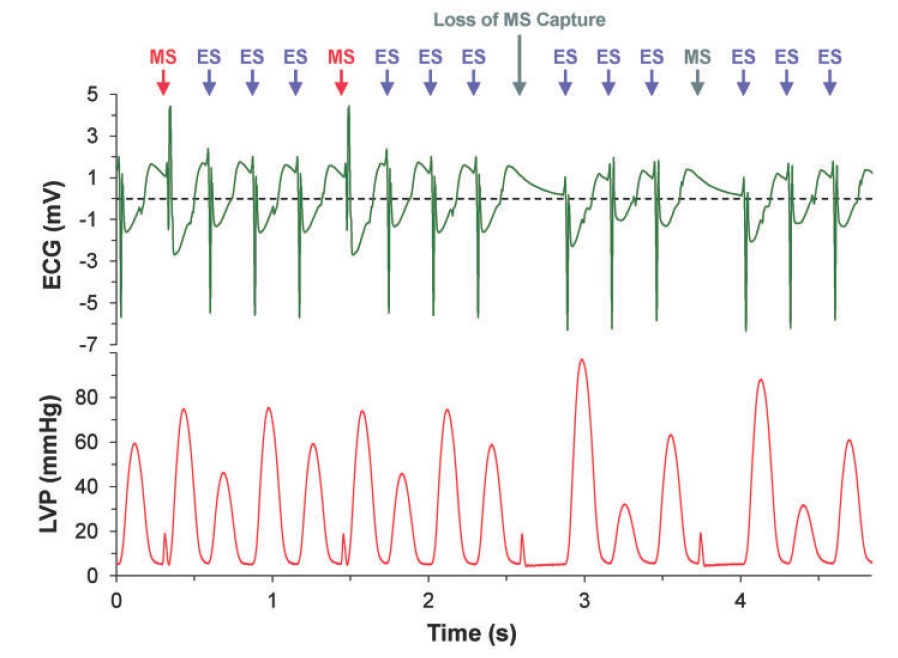
Fig.2 Electrocardiogram (ECG) and left ventricular pressure (LVP) recordings during a train of 1:3 mechanical: electrical (MS:ES) stimulations at a total stimulation rate of 3 Hz during which loss of MS capture occurs after 2.5 s, while ES capture is maintained.
Highlights
Isolated heart perfused with increasing concentrations of test article
Spontaneously-beating or stimulated hearts
Dissection and compilation of RR, PR, QRS, QT, QTc, intervals and LVP, dLVP/dt
Continuous monophasic action potential (MAP), coronary pressure and flow monitoring
Left-ventricle contraction
Rat, guinea-pig, rabbit, dog hearts
Pre-IND (GLP-compliant) or exploratory designs (screen = non-GLP)
References
Liao R, et al. The continuing evolution of the Langendorff and ejecting murine heart: new advances in cardiac phenotyping. AJP Hear Circ Physiol. 2012; 303: H156–H167.
Quinn TA, Kohl P. Comparing maximum rate and sustainability of pacing by mechanical vs. electrical stimulation in the Langendorff-perfused rabbit heart. EP Eur. 2016; 18: 85-93.
Related Section
Inquiry

